Electrons are not merely passive bystanders in the universe; they are the fundamental architects of nearly every chemical transformation and technological marvel. They orchestrate the flow of energy, forge the bonds that hold matter together, and dictate the very essence of electrical conductivity. This dual role makes them the bedrock upon which both sophisticated chemical synthesis and the intricate tapestry of modern electronics are woven. In the realm of chemistry, electrons are the key players in redox reactions, the linchpin of bond formation, and the driving force behind catalytic activity. In technology, the ability to meticulously manage the movement and interactions of electrons is the silent engine powering everything from the microscopic pathways of electronic circuits and the complex algorithms of artificial intelligence to the energy-harvesting mechanisms of solar cells and the mind-bending potential of quantum computers. Historically, electrons have been largely confined to the atomic orbitals, a limitation that has inherently restricted their full exploitative potential. However, the emergence of materials known as electrides, where electrons exhibit independent mobility, has dramatically expanded the horizons, unlocking the door to a cascade of remarkable new capabilities.
"By mastering the art of controlling these mobile electrons, we are empowered to design and engineer materials that can perform feats far beyond what nature, in its current state, has envisioned," explains Dr. Evangelos Miliordos, a distinguished Associate Professor of Chemistry at Auburn University and the senior architect of this groundbreaking study. His team’s work, underpinned by sophisticated and cutting-edge computational modeling, represents a significant leap forward in our understanding and manipulation of electron behavior.
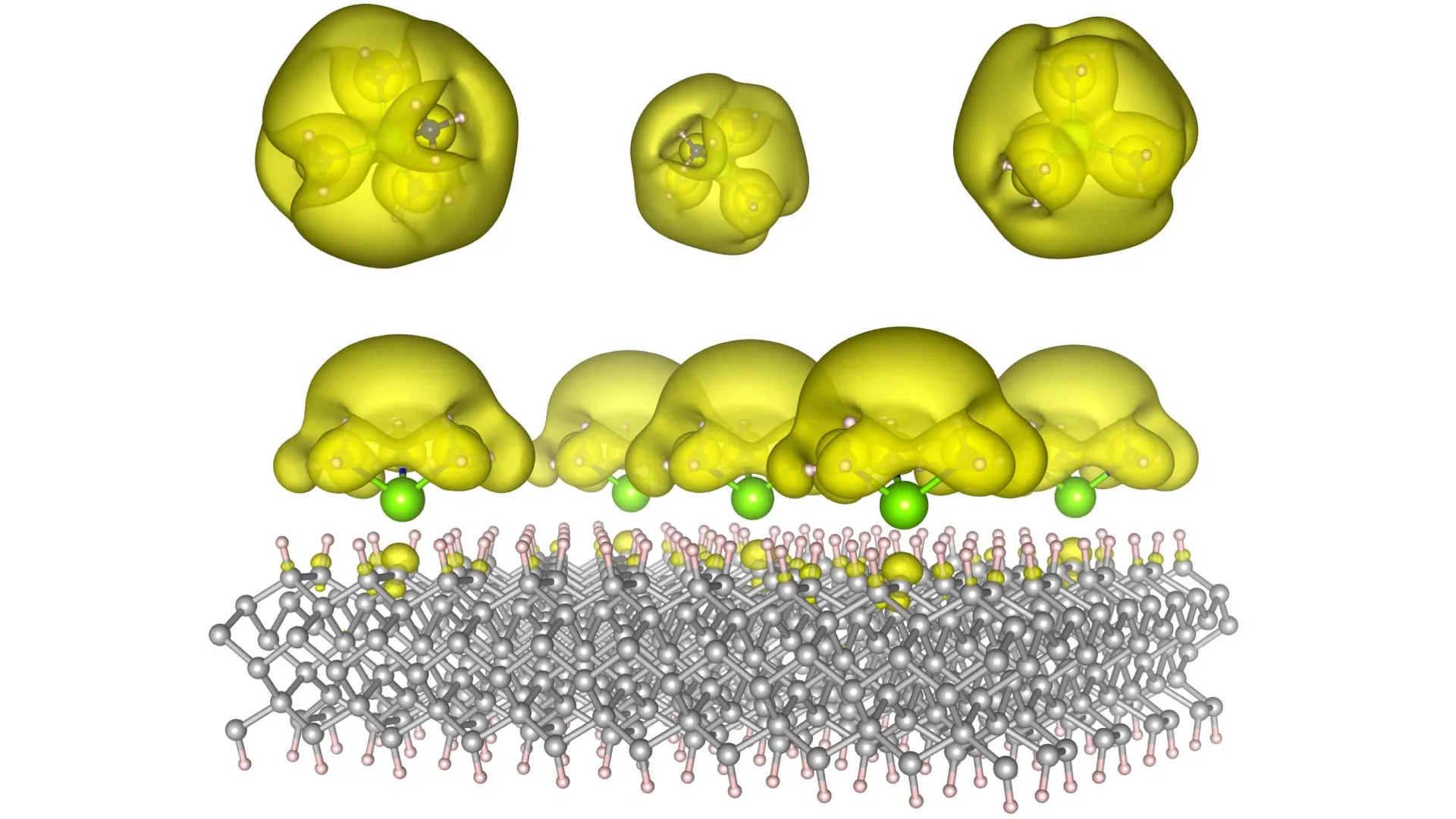
To realize this ambitious goal, the Auburn research collective devised and fabricated innovative material architectures, christened "Surface Immobilized Electrides." This ingenious approach involves the precise attachment of solvated electron precursors to exceptionally stable surfaces, such as the robust framework of diamond and the versatile substrate of silicon carbide. This strategic configuration bestows upon the electronic characteristics of these electrides a remarkable combination of durability and tunability. By subtly altering the spatial arrangement and intermolecular distances of the constituent molecules, the researchers can precisely dictate the electron distribution. This allows for electrons to either coalesce into discrete, isolated "islands," which can function as the fundamental building blocks – or qubits – for the next generation of advanced computing, or to spread out and form extensive, interconnected "seas" that significantly enhance and promote complex chemical reactions.
This inherent versatility is precisely what imbues this discovery with its truly transformative potential. One manifestation of this innovation could pave the way for the development of immensely powerful quantum computers, capable of tackling computational problems that currently lie far beyond the reach of even the most advanced supercomputers. Another avenue of application could lay the foundation for revolutionary catalysts, meticulously engineered to accelerate essential chemical reactions with unparalleled efficiency. This, in turn, could profoundly reshape the methodologies for producing vital commodities such as fuels, life-saving pharmaceuticals, and a vast array of industrial materials.
"As our global society relentlessly pushes the boundaries of current technological capabilities, the demand for novel and advanced materials is experiencing an exponential surge," observes Dr. Marcelo Kuroda, an equally distinguished Associate Professor of Physics at Auburn. "Our research unequivocally demonstrates a novel and promising pathway towards the creation of materials that not only offer profound opportunities for fundamental investigations into the intricate interactions within matter but also possess the capacity for significant practical applications."
Previous attempts to harness the power of electrides were often hampered by their inherent instability and the significant challenges associated with scaling up their production. By ingeniously depositing these electron-rich precursors directly onto solid surfaces, the Auburn team has successfully circumvented these formidable barriers. They have proposed a compelling and versatile family of material structures that hold the promise of transitioning from theoretical constructs and laboratory curiosities to tangible, real-world devices. "While this work is rooted in fundamental scientific inquiry, its implications are profoundly real and far-reaching," asserts Dr. Konstantin Klyukin, an Assistant Professor of Materials Engineering at Auburn, emphasizing the tangible impact of their theoretical breakthroughs. "We are on the cusp of realizing technologies that possess the potential to fundamentally alter how we compute and, equally importantly, how we manufacture the goods that define our modern world."
The intellectual genesis and execution of this pioneering theoretical study involved a synergistic collaboration of faculty members from diverse disciplines across Auburn University, including chemistry, physics, and materials engineering. "This is merely the nascent stage of an extraordinary journey," Dr. Miliordos adds with evident enthusiasm, reflecting on the vast uncharted territory that lies ahead. "By diligently learning how to harness and precisely control the behavior of these free electrons, we can realistically envision a future characterized by computers of unprecedented speed, intelligent machines of remarkable adaptability, and the emergence of entirely new technologies that, at this very moment, remain beyond our wildest imaginings."
The comprehensive scientific paper detailing these groundbreaking discoveries, provocatively titled "Electrides with Tunable Electron Delocalization for Applications in Quantum Computing and Catalysis," was also graced with the co-authorship of talented graduate students Andrei Evdokimov and Valentina Nesterova, whose contributions were instrumental to the study’s success. The research was generously supported by vital funding from the U.S. National Science Foundation, alongside the indispensable computational resources provided by Auburn University.