Electrons are the unsung heroes of the universe, orchestrating virtually every chemical and technological process we rely upon. They are the architects of energy transfer, the glue of chemical bonds, and the very essence of electrical conductivity. From the intricate dance of atoms in chemical synthesis to the sophisticated circuits powering our digital world, electrons are the fundamental currency. In the realm of chemistry, they drive redox reactions, forge new molecular bonds, and act as catalysts, accelerating the creation of countless substances. In technology, the ability to precisely govern electron movement and interaction forms the bedrock of everything from the microprocessors in our laptops and the algorithms powering artificial intelligence, to the photovoltaic cells capturing solar energy and the enigmatic promise of quantum computers. Traditionally, electrons are bound tightly to individual atoms, a constraint that has historically limited their potential applications. However, a special class of materials, known as electrides, defies this norm. In electrides, electrons exist in a delocalized state, moving freely and independently, thereby paving the way for a breathtaking array of novel capabilities.
Dr. Evangelos Miliordos, Associate Professor of Chemistry at Auburn and the senior author of the study, articulated the profound implications of this discovery. "By learning how to control these free electrons, we can design materials that do things nature never intended," he explained, highlighting the study’s foundation in advanced computational modeling. This mastery over free electrons is not merely an academic pursuit; it represents a paradigm shift in materials science, enabling the creation of substances with properties previously confined to theoretical speculation.
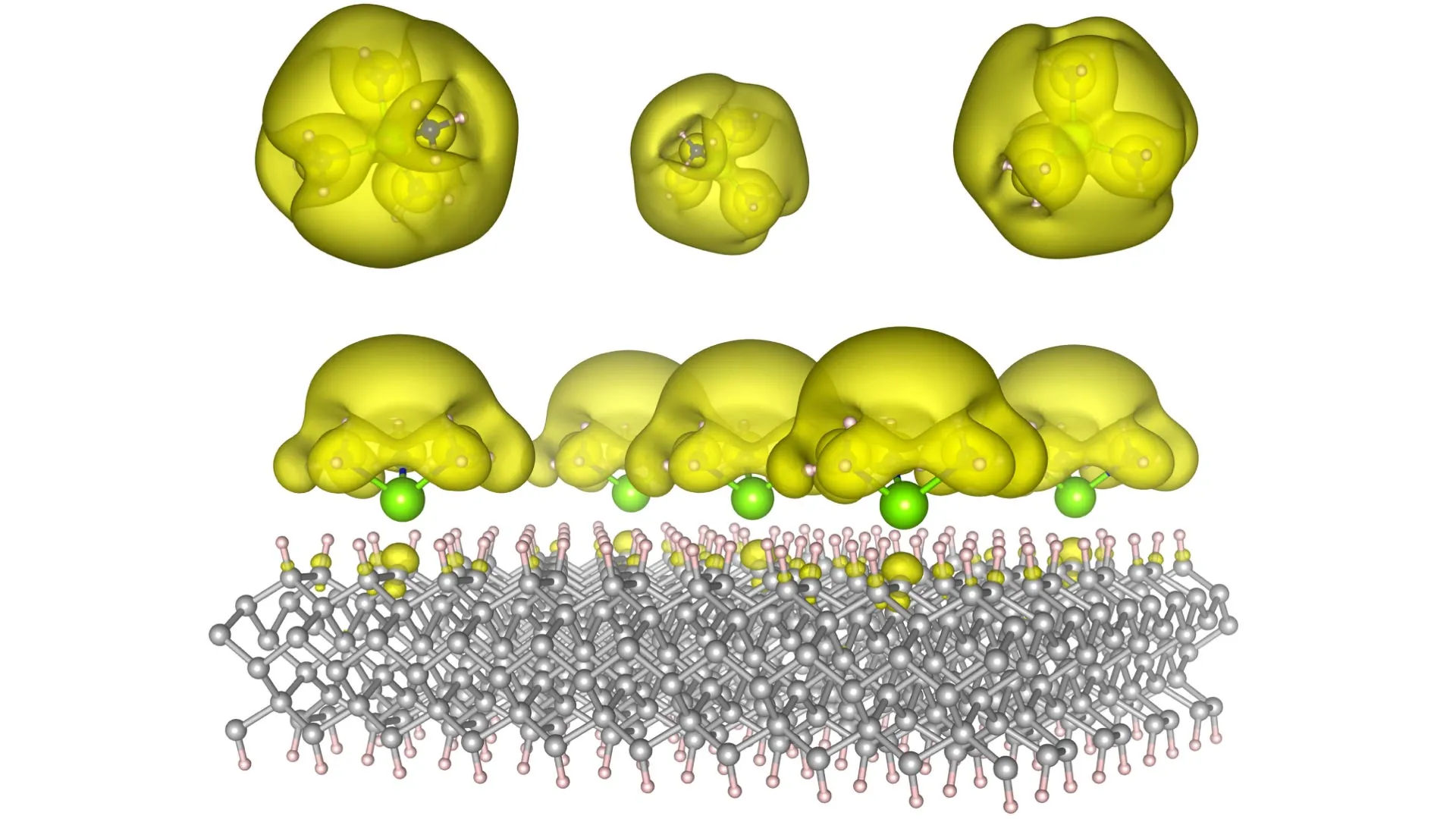
The breakthrough achieved by the Auburn team involves the ingenious creation of novel material architectures, dubbed Surface Immobilized Electrides. This innovative approach entails anchoring solvated electron precursors onto robust and stable surfaces, such as diamond and silicon carbide. This strategic immobilization imbues the electrides with a dual benefit: their electronic characteristics become both remarkably durable and exquisitely tunable. The key lies in the precise arrangement of the molecules. By subtly altering this molecular architecture, researchers can dictate the spatial distribution of electrons. They can be coaxed to cluster into discrete, isolated "islands," each behaving as an independent quantum bit, or qubit – the fundamental unit of information in quantum computing. Alternatively, these electrons can be encouraged to spread out into vast, interconnected "seas," creating environments that are exceptionally conducive to facilitating complex and rapid chemical reactions.
This remarkable versatility is precisely what bestows upon this discovery its transformative potential. In one manifestation, these engineered materials could serve as the foundational building blocks for a new generation of quantum computers. Such machines, empowered by the unique properties of quantum mechanics, would possess the capacity to tackle problems that are currently intractable for even the most powerful supercomputers, opening doors to breakthroughs in fields like drug discovery, materials science, and complex system modeling. In another application, these materials could form the basis of cutting-edge catalysts. These catalysts would possess the ability to dramatically accelerate essential chemical reactions, potentially revolutionizing industries ranging from energy production, by enabling more efficient fuel synthesis, to pharmaceuticals and the manufacturing of advanced industrial materials, leading to more sustainable and cost-effective production processes.
Dr. Marcelo Kuroda, Associate Professor of Physics at Auburn, underscored the growing demand driving this research. "As our society pushes the limits of current technology, the demand for new kinds of materials is exploding," he stated. "Our work shows a new path to materials that offer both opportunities for fundamental investigations on interactions in matter as well as practical applications." This sentiment reflects the dual nature of the discovery: it not only deepens our fundamental understanding of electron behavior but also offers tangible pathways to solve pressing technological challenges.
The historical development of electrides has been hampered by their inherent instability and the significant challenges associated with scaling their production. By ingeniously depositing these electron-rich species directly onto solid surfaces, the Auburn team has effectively circumvented these long-standing obstacles. They have proposed a new family of material structures that can transition from the realm of theoretical models to the tangible reality of functional devices. "This is fundamental science, but it has very real implications," emphasized Dr. Konstantin Klyukin, Assistant Professor of Materials Engineering at Auburn. "We’re talking about technologies that could change the way we compute and the way we manufacture." This assertion points to the broad societal impact of the research, touching upon two of the most critical sectors of the modern economy.
The theoretical underpinnings of this groundbreaking research were spearheaded by a collaborative effort involving faculty from Auburn University’s departments of chemistry, physics, and materials engineering. This interdisciplinary approach was crucial in tackling the multifaceted challenges associated with understanding and manipulating electron behavior at such a fundamental level. "This is just the beginning," Dr. Miliordos added optimistically. "By learning how to tame free electrons, we can imagine a future with faster computers, smarter machines, and new technologies we haven’t even dreamed of yet." This forward-looking statement encapsulates the boundless potential inherent in controlling free electrons, suggesting a future replete with innovations that could redefine human capabilities.
The comprehensive study, titled "Electrides with Tunable Electron Delocalization for Applications in Quantum Computing and Catalysis," was further enriched by the contributions of graduate students Andrei Evdokimov and Valentina Nesterova. The research received vital support from the U.S. National Science Foundation, providing essential funding for the scientific inquiry, and from Auburn University’s robust computing resources, which were indispensable for the advanced computational modeling that formed the bedrock of the discovery. This collaborative ecosystem, combining fundamental research, theoretical modeling, and dedicated student involvement, exemplifies the power of academic innovation in pushing the boundaries of scientific knowledge and technological possibility. The implications of this research extend far beyond the laboratory, hinting at a future where materials are not merely discovered but intelligently designed to meet the ever-evolving demands of a technologically driven world. The ability to precisely engineer the quantum behavior of electrons within materials represents a profound leap forward, promising to redefine the very fabric of our technological landscape.