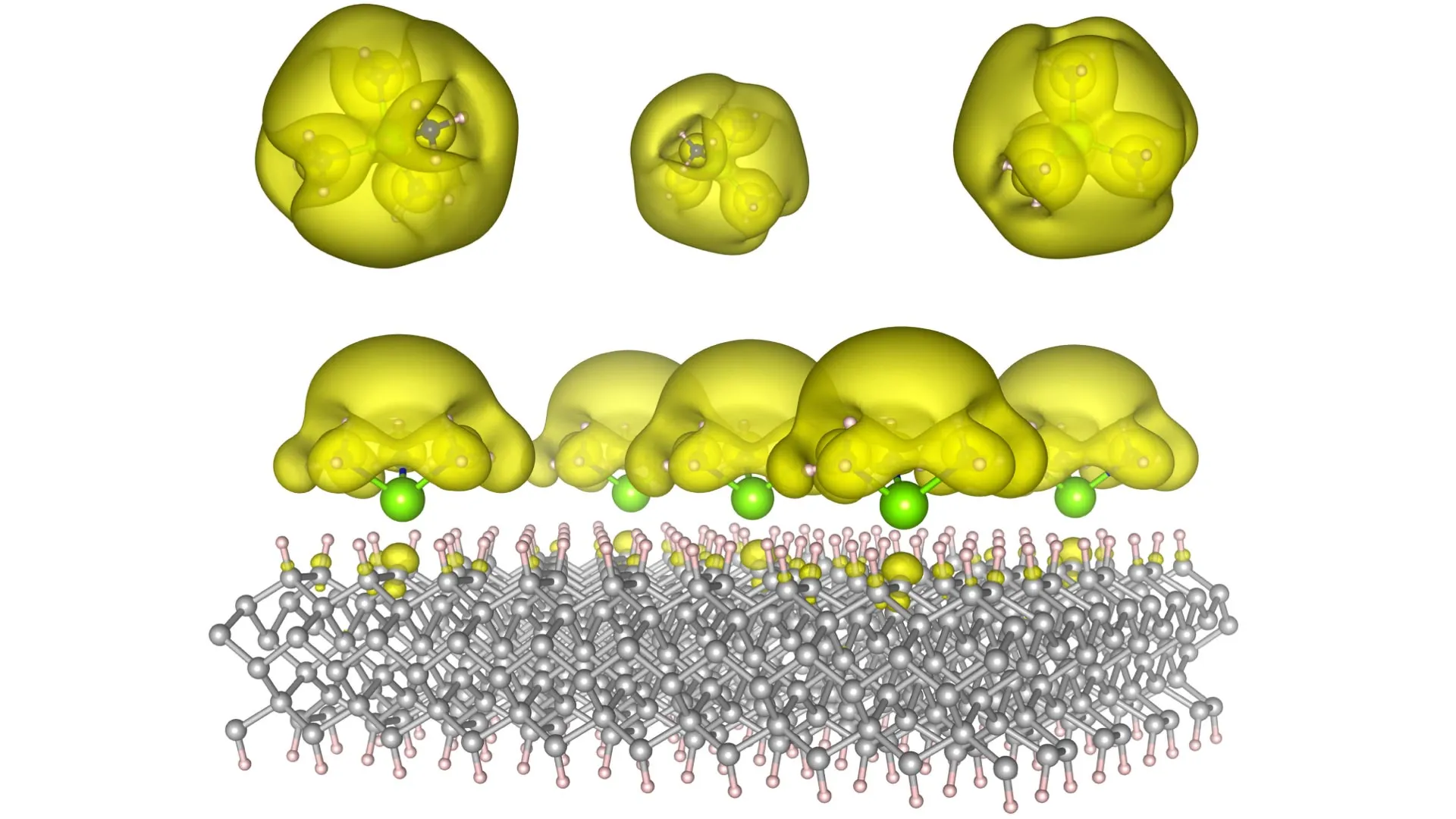
Electrons, the ubiquitous carriers of negative charge, are the very bedrock upon which nearly every conceivable chemical reaction and technological advancement is built. Their dynamic interplay is the engine driving energy transfer, forging the bonds that hold matter together, and dictating the flow of electricity. From the fundamental processes of chemical synthesis to the sophisticated architecture of modern electronics, the management of electron movement and interaction is paramount. In the intricate dance of chemical reactions, electrons are the essential agents enabling redox processes, the formation of new chemical bonds, and the catalytic activity that accelerates transformations. In the realm of technology, the ability to control how electrons move, interact, and are localized or delocalized underpins everything from the intricate pathways of electronic circuits and the learning algorithms of artificial intelligence systems to the energy harvesting mechanisms of solar cells and the mind-bending potential of quantum computers. Traditionally, electrons are bound tightly to individual atoms, a constraint that inherently limits their functional potential. However, a special class of materials known as electrides shatters these limitations, featuring electrons that move independently, thus unlocking a vista of extraordinary new capabilities and applications.
Dr. Evangelos Miliordos, an Associate Professor of Chemistry at Auburn University and the senior architect of this groundbreaking study, which was significantly bolstered by sophisticated computational modeling, articulates the profound implications of this discovery. "By mastering the art of controlling these free-roaming electrons," he explains, "we are empowered to engineer materials that can perform functions far beyond what nature has historically dictated."
In pursuit of this ambitious goal, the dedicated team at Auburn has ingeniously engineered novel material architectures, christened Surface Immobilized Electrides. This innovation involves the strategic attachment of solvated electron precursors to robust and stable surfaces, such as those offered by diamond and silicon carbide. This ingenious configuration bestows upon the electronic characteristics of these electrides a dual advantage: remarkable durability and exceptional tunability. The precise arrangement of the molecular components within these structures dictates the behavior of the electrons. By subtly altering this molecular architecture, scientists can orchestrate the electrons to either coalesce into isolated "islands," akin to the fundamental building blocks of quantum bits essential for advanced computing, or to disperse widely into expansive "seas," thereby fostering and accelerating complex chemical reactions.
It is precisely this inherent versatility that imbues the discovery with its truly transformative potential. One manifestation of this breakthrough could pave the way for the development of immensely powerful quantum computers, machines capable of tackling computational problems that currently lie far beyond the reach of even the most advanced supercomputers. Concurrently, another facet of this discovery could serve as the foundational element for the creation of cutting-edge catalysts. These catalysts would possess the remarkable ability to significantly accelerate crucial chemical reactions, potentially revolutionizing the production of essential commodities such as fuels, life-saving pharmaceuticals, and a vast array of industrial materials.
Dr. Marcelo Kuroda, another Associate Professor of Physics at Auburn, emphasizes the pressing need for novel materials in our rapidly advancing technological landscape. "As our society relentlessly pushes the boundaries of current technological capabilities, the demand for new and innovative types of materials is experiencing an explosive surge," he observes. "Our research not only unveils a novel pathway to developing these advanced materials but also presents both invaluable opportunities for fundamental investigations into the complex interactions within matter and a clear roadmap for practical, real-world applications."
Previous iterations of electride materials were notoriously unstable and presented significant challenges in terms of scalability, hindering their transition from laboratory curiosities to practical applications. By ingeniously depositing these electrides directly onto solid surfaces, the Auburn team has effectively surmounted these critical barriers. They have put forth a compelling vision for an entire family of material structures that possess the potential to evolve from theoretical constructs into tangible, functional devices. Dr. Konstantin Klyukin, an Assistant Professor of Materials Engineering at Auburn, underscores the profound impact of this fundamental scientific endeavor. "While this work is rooted in fundamental science," he states, "its implications are exceptionally tangible. We are on the cusp of developing technologies that have the capacity to fundamentally alter the way we compute and the way we manufacture goods."
The theoretical underpinnings of this groundbreaking study were spearheaded by a collaborative effort involving esteemed faculty members from the disciplines of chemistry, physics, and materials engineering at Auburn University. Dr. Miliordos concludes with an optimistic outlook on the future. "This is merely the nascent stage of what promises to be a revolutionary journey," he asserts. "By unlocking the secrets of taming free electrons, we can vividly envision a future characterized by exponentially faster computers, more intelligent and adaptive machines, and the emergence of entirely new technologies that, at present, we can only begin to dream of."
The seminal study, provocatively titled "Electrides with Tunable Electron Delocalization for Applications in Quantum Computing and Catalysis," also bore the significant contributions of graduate students Andrei Evdokimov and Valentina Nesterova. This ambitious research endeavor received crucial support from the U.S. National Science Foundation and was facilitated by the extensive computing resources available at Auburn University. The implications of this research are far-reaching, suggesting a paradigm shift in how we conceive of and engineer materials for the next generation of technological innovation, from unlocking the mysteries of the universe with advanced computation to sustainably producing the resources that underpin modern civilization.