Electrons, the tiny, negatively charged particles, are the unsung heroes of virtually every chemical reaction and technological process. Their dynamic interactions govern energy transfer, the formation of chemical bonds, and the flow of electricity, forming the bedrock upon which both complex chemical syntheses and the intricate circuitry of modern electronics are built. In the realm of chemistry, electrons are indispensable for redox reactions, the formation of molecular bonds, and the catalytic activity that accelerates transformations. In technology, the ability to manage and manipulate electron movement and interaction is what underpins the functionality of everything from the microprocessors in our laptops and the sophisticated algorithms driving artificial intelligence to the efficiency of solar cells and the nascent power of quantum computers. Traditionally, electrons have been largely confined to orbits around atomic nuclei, a limitation that has inherently restricted their potential applications. However, the emergence of materials known as electrides, where electrons are not tethered to specific atoms but instead exist in a delocalized state, has begun to shatter these constraints, heralding the possibility of remarkable new capabilities.
"By unlocking the secrets to controlling these free-moving electrons, we are empowered to design and engineer materials with functionalities that far exceed what nature has conventionally provided," explains Dr. Evangelos Miliordos, an Associate Professor of Chemistry at Auburn University and the senior author of the groundbreaking study. The research, which leveraged sophisticated computational modeling techniques, represents a significant leap forward in our understanding and manipulation of electronic behavior in materials.
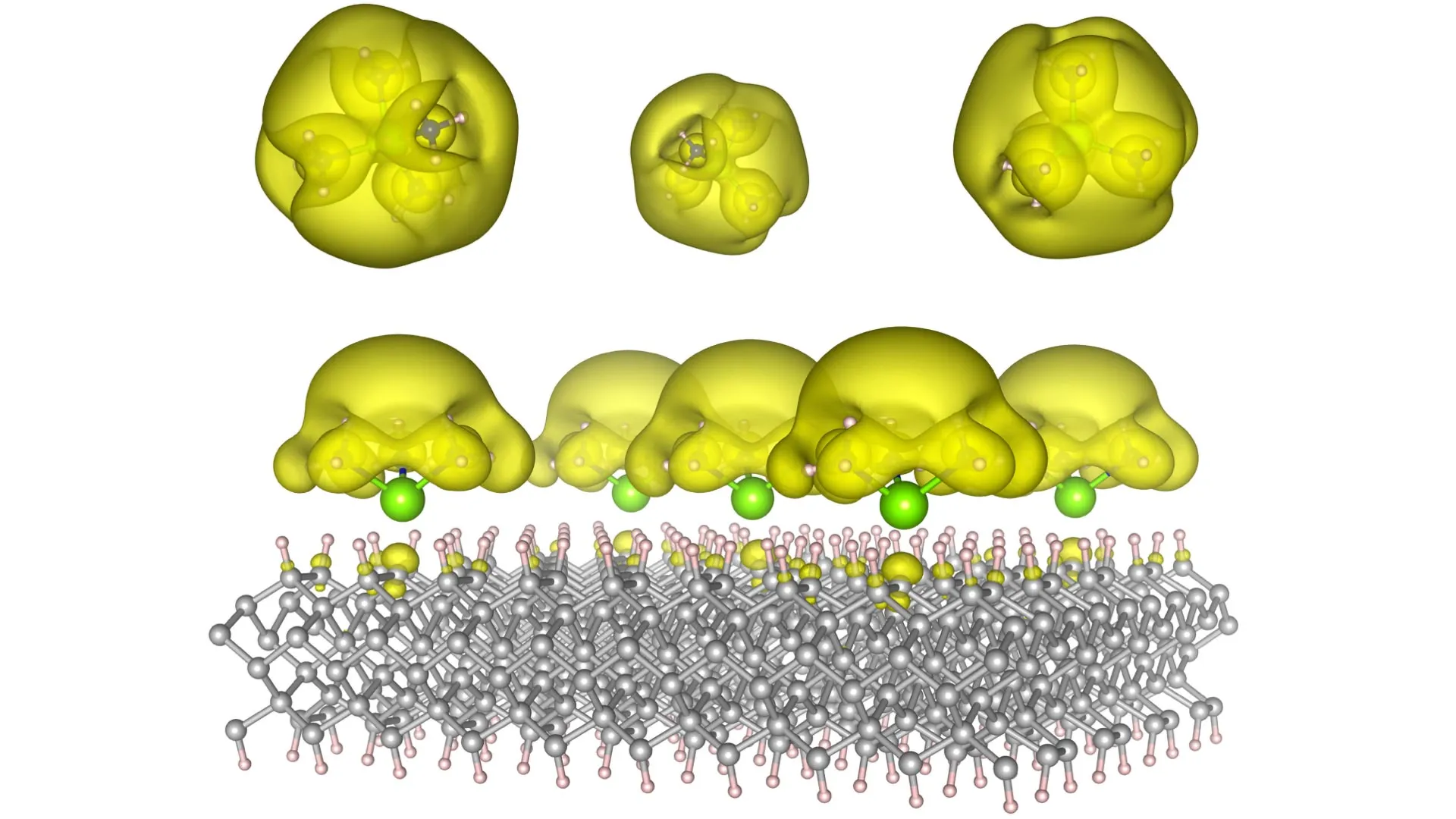
The innovative approach taken by the Auburn team involved the creation of unique material architectures they call Surface Immobilized Electrides. This was achieved by strategically attaching solvated electron precursors, which are molecules that readily release electrons, to robust and stable surfaces such as diamond and silicon carbide. This clever immobilization strategy imbues the electrides with remarkable durability and, crucially, allows for their electronic characteristics to be precisely adjusted. By subtly altering the arrangement and spacing of the molecular complexes on the surface, the researchers can dictate whether the electrons tend to cluster into isolated "islands" or spread out into more extended "seas."
This exquisite tunability is the cornerstone of the discovery’s transformative potential. When electrons are encouraged to form isolated "islands," these islands can function analogously to quantum bits, or qubits, the fundamental units of information in quantum computing. This could pave the way for the development of powerful quantum computers capable of tackling problems that are currently intractable for even the most advanced supercomputers, such as complex molecular simulations for drug discovery or the optimization of intricate logistical networks. Conversely, when electrons are induced to spread into extended "seas," this delocalized state can significantly promote and accelerate complex chemical reactions. This has immense implications for the field of catalysis, potentially revolutionizing the production of essential chemicals, fuels, pharmaceuticals, and industrial materials by enabling faster, more efficient, and more sustainable synthesis pathways.
"As our global society continues to push the boundaries of existing technological capabilities, the demand for novel and advanced materials is experiencing an exponential surge," observes Dr. Marcelo Kuroda, an Associate Professor of Physics at Auburn University and a key contributor to the research. "Our work illuminates a new and promising avenue for the development of materials that not only offer profound opportunities for fundamental scientific investigations into the intricate interactions within matter but also possess immense practical applicability across a wide spectrum of industries."
Historically, earlier iterations of electride materials have presented significant challenges, often characterized by their instability and difficulties in scaling up production for practical applications. By directly depositing these electron-rich precursors onto solid surfaces, the Auburn team has effectively surmounted these critical barriers. Their research proposes a versatile family of material structures that can transition from theoretical concepts to tangible, real-world devices. "While this research is rooted in fundamental scientific inquiry, its implications are exceptionally tangible and far-reaching," emphasizes Dr. Konstantin Klyukin, an Assistant Professor of Materials Engineering at Auburn University. "We are envisioning the development of technologies that have the potential to fundamentally alter how we approach computation and how we manufacture goods on an industrial scale."
The theoretical underpinnings of this groundbreaking study were a testament to interdisciplinary collaboration, with faculty members from Auburn University’s departments of Chemistry, Physics, and Materials Engineering contributing their expertise. "This represents merely the nascent stages of what we believe will be a profoundly impactful field of research," Dr. Miliordos adds with palpable enthusiasm. "By diligently learning how to effectively ‘tame’ and control these free electrons, we can begin to conceptualize a future characterized by significantly faster and more powerful computers, increasingly intelligent machines capable of complex problem-solving, and the realization of entirely new technologies that, at this moment, we can scarcely even dream of."
The comprehensive study, titled "Electrides with Tunable Electron Delocalization for Applications in Quantum Computing and Catalysis," was further enriched by the dedicated contributions of graduate students Andrei Evdokimov and Valentina Nesterova, who played vital roles in the research and data analysis. The project received crucial support from the U.S. National Science Foundation, recognizing its potential scientific significance, and benefited from the extensive computational resources provided by Auburn University, which were essential for conducting the advanced modeling and simulations that underpinned the discovery. This pioneering work not only expands the fundamental understanding of electron behavior in materials but also lays the groundwork for a future where quantum crystals and precisely engineered electrides drive the next wave of technological innovation, reshaping industries and redefining the limits of what is possible.