Electrons are the silent architects of nearly every chemical reaction and technological marvel we encounter. They are the linchpins of energy transfer, the force behind chemical bonding, and the very essence of electrical conductivity. From the delicate dance of atoms in chemical synthesis to the sophisticated circuitry powering our modern electronics, the behavior of electrons is paramount. In the realm of chemistry, electrons facilitate vital processes such as redox reactions, the formation of new bonds, and catalytic activity, which can accelerate reactions dramatically. In technology, understanding and controlling electron movement and interaction is the bedrock of everything from the intricate pathways of electronic circuits and the adaptive learning of artificial intelligence systems to the efficient energy conversion of solar cells and the mind-bending capabilities of quantum computers. Traditionally, electrons are bound to specific atoms, a confinement that has historically limited their diverse applications. However, a special category of materials called electrides defies this norm. In electrides, electrons are not tethered to individual atoms but exist as mobile entities, paving the way for astonishing new functionalities and technological breakthroughs.
"By mastering the art of controlling these free-moving electrons, we gain the power to engineer materials that can perform tasks far beyond the capabilities envisioned by nature itself," proclaims Dr. Evangelos Miliordos, an Associate Professor of Chemistry at Auburn University and the senior author of this pivotal study. This pioneering work, built upon sophisticated computational modeling, represents a significant leap forward in our understanding and manipulation of electron behavior.
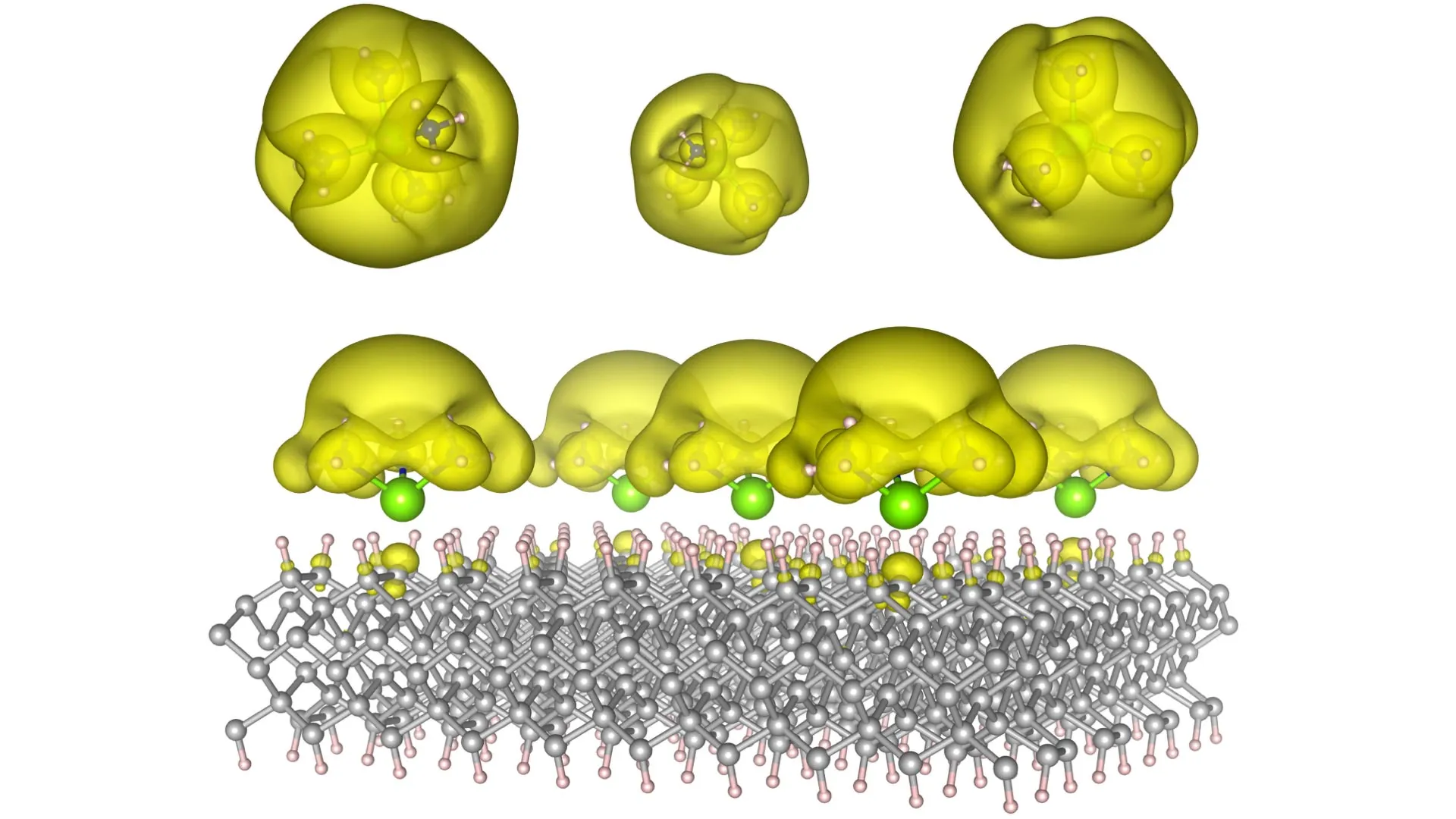
The Auburn team’s ingenious approach involved the creation of novel material architectures, dubbed Surface Immobilized Electrides. This was achieved by anchoring solvated electron precursors onto exceptionally stable surfaces, such as diamond and silicon carbide. This strategic immobilization imparts a dual benefit to the electrides: their electronic characteristics become both remarkably durable and exquisitely tunable. The key to this versatility lies in the ability to precisely alter the arrangement of the molecular components. By judiciously modifying this molecular architecture, researchers can orchestrate the electrons to either coalesce into distinct, isolated "islands" – behaving like sophisticated quantum bits crucial for advanced computing – or to spread out into expansive, interconnected "seas" that can powerfully facilitate complex chemical transformations.
It is this extraordinary versatility that imbues the discovery with its truly transformative potential. One manifestation of this breakthrough could be the development of immensely powerful quantum computers. These machines would possess the computational prowess to tackle problems that are currently intractable for even the most advanced supercomputers, unlocking solutions to challenges in fields like drug discovery, climate modeling, and materials science. Alternatively, another configuration of these materials could serve as the foundation for cutting-edge catalysts. These catalysts would possess the ability to dramatically accelerate essential chemical reactions, potentially revolutionizing the production of fuels, pharmaceuticals, and a vast array of industrial materials, leading to more sustainable and efficient manufacturing processes.
"As our global society consistently pushes the boundaries of current technological capabilities, the demand for novel and innovative materials is experiencing an unprecedented surge," observes Dr. Marcelo Kuroda, an Associate Professor of Physics at Auburn. "Our research illuminates a promising new pathway toward the creation of materials that not only offer profound opportunities for fundamental investigations into the intricate interactions within matter but also hold immense promise for practical, real-world applications."
Previous iterations of electrides were often plagued by inherent instability and significant challenges in scaling up production. By ingeniously depositing these electron-rich precursors directly onto robust solid surfaces, the Auburn team has effectively surmounted these critical barriers. They have put forth a compelling proposal for a new family of material structures that can realistically transition from theoretical models to tangible, functional devices. "While this research is rooted in fundamental scientific principles, its implications are profoundly practical," emphasizes Dr. Konstantin Klyukin, an Assistant Professor of Materials Engineering at Auburn. "We are on the cusp of developing technologies that have the potential to fundamentally reshape how we approach computation and how we undertake manufacturing processes."
The theoretical underpinnings of this transformative study were meticulously developed through the collaborative efforts of faculty members spanning the disciplines of chemistry, physics, and materials engineering at Auburn University. "This is merely the nascent stage of what we believe will be a revolutionary journey," Dr. Miliordos adds with evident enthusiasm. "By diligently pursuing our understanding and mastery of free electrons, we can realistically envision a future characterized by computers that operate at speeds we can only currently dream of, intelligent machines that possess unprecedented adaptability, and entirely new technological paradigms that are currently beyond our wildest imaginations."
The seminal study, aptly titled "Electrides with Tunable Electron Delocalization for Applications in Quantum Computing and Catalysis," also featured the significant contributions of graduate students Andrei Evdokimov and Valentina Nesterova as co-authors. The research was generously supported by critical funding from the U.S. National Science Foundation, alongside essential computing resources provided by Auburn University, underscoring the collaborative and well-supported nature of this pioneering scientific endeavor. This breakthrough represents not just an incremental improvement in material science but a paradigm shift that promises to redefine the landscape of technology for generations to come.