The fundamental obstacle preventing the direct injection of antibody drugs has historically been their inherent low concentration. To achieve a therapeutically effective dose, a substantial volume of liquid is required. Attempting to simply increase the concentration of antibodies in a solution to reduce the required volume for injection runs into a critical physical limitation: the viscosity of the solution increases dramatically. At the high concentrations needed to fit a full dose into the small volume of a standard syringe, these antibody solutions become excessively thick, making them impossible to inject through the narrow gauge needles typically used for subcutaneous administration. This rheological challenge has been a persistent hurdle for researchers and pharmaceutical developers aiming to transition antibody therapies from the clinic to the home.
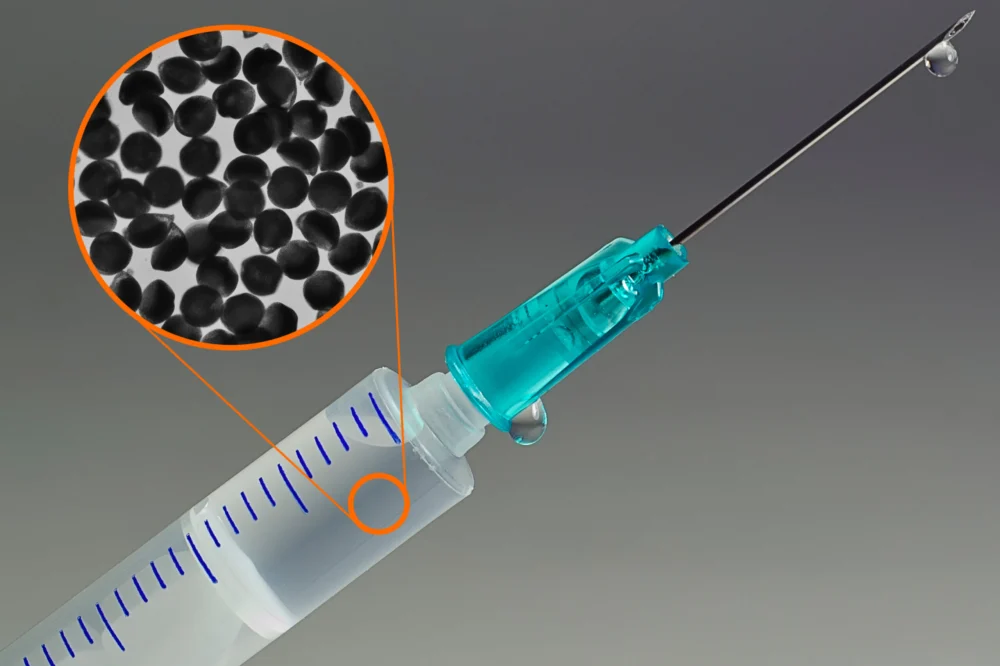
In 2023, Doyle’s lab made a substantial stride toward overcoming this viscosity barrier by pioneering a technique to generate highly concentrated antibody formulations. Their innovative approach involved encapsulating antibodies within microscopic hydrogel particles. These hydrogel matrices acted as tiny reservoirs, capable of holding a significantly higher density of antibodies than a free solution. While this method proved effective in achieving high concentrations, it relied on a centrifugation process. Centrifugation, while a standard laboratory technique, poses significant challenges when it comes to scaling up for industrial manufacturing. The energy requirements, specialized equipment, and time involved in large-scale centrifugation can be prohibitive, limiting the commercial viability of therapies produced using this method. Therefore, while a crucial proof-of-concept, the 2023 development pointed towards a need for a more manufacturing-friendly approach.
The latest research, detailed in a new study published in Advanced Materials, represents a significant evolution of their earlier work, introducing a fundamentally different and more scalable manufacturing strategy. Instead of relying on centrifugation, the MIT team has ingeniously employed a microfluidic setup. This advanced technology allows for precise control over fluid manipulation at the microscale. The core of their new process involves creating tiny droplets containing antibodies dissolved in a watery prepolymer solution. These antibody-laden droplets are then suspended within an immiscible organic solvent. The key step involves carefully dehydrating these droplets. As water evaporates, the antibodies become highly concentrated and solidify within a hydrogel matrix, forming stable, solid antibody particles. Following this dehydration and solidification process, the organic solvent is removed and replaced with a fresh aqueous solution, rendering the concentrated antibodies rehydratable and ready for administration in a form suitable for injection. This microfluidic approach offers a significant advantage in terms of scalability, as microfluidic systems are more amenable to continuous processing and large-scale production compared to batch centrifugation methods.
The efficacy of this new formulation technique was rigorously demonstrated. The researchers successfully created semi-solid antibody particles approximately 100 microns in diameter. Crucially, they then tested the injectability of these concentrated formulations. The results were highly encouraging: the force required to depress the plunger of a standard syringe containing the reformulated antibody solution was measured to be less than 20 newtons. Talia Zheng, an MIT graduate student and the lead author of the new study, emphasized the significance of this finding, stating, "That is less than half of the maximum acceptable force that people usually try to aim for." This benchmark is critical because it aligns with established ergonomic guidelines for manual injection, ensuring that the process is comfortable and manageable for patients and healthcare providers alike, avoiding the discomfort and difficulty associated with injecting highly viscous fluids.
The practical implications of this breakthrough are substantial. The team demonstrated that their reformulated particles could deliver a significant therapeutic load in a minimal volume. Specifically, they showed that more than 700 milligrams of antibody – an amount sufficient for most therapeutic applications – could be administered in a single dose using a standard two-milliliter syringe. This represents a dramatic reduction in injection volume compared to current IV formulations. Furthermore, the stability of these novel antibody formulations was rigorously assessed. Under standard refrigeration conditions, the particles remained stable for a minimum of four months, a crucial factor for ensuring the shelf life and reliability of pharmaceutical products. Looking ahead, the researchers have ambitious plans to translate this laboratory success into tangible clinical benefits. Their next steps involve conducting animal studies to evaluate the safety and efficacy of these injectable antibody formulations in a living system. Concurrently, they are actively working on optimizing and scaling up the manufacturing process to pave the way for potential clinical trials and eventual commercialization. This innovative approach by Professor Doyle and his team promises to usher in a new era of antibody therapy, making these life-saving treatments more accessible, convenient, and patient-friendly than ever before. The ability to administer potent antibody therapies via simple injection could dramatically improve treatment adherence, reduce healthcare burdens, and ultimately enhance the quality of life for millions of patients worldwide. The development signifies a paradigm shift from hospital-bound infusions to accessible, at-home or clinic-based injections, democratizing access to advanced biological therapies.