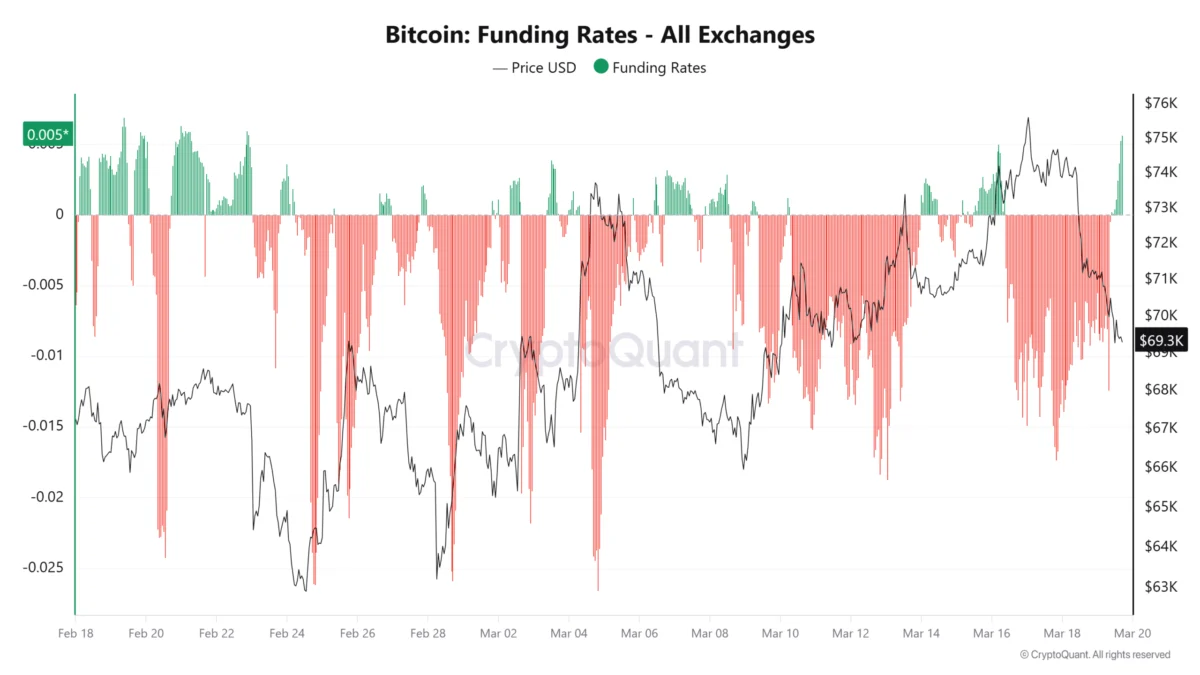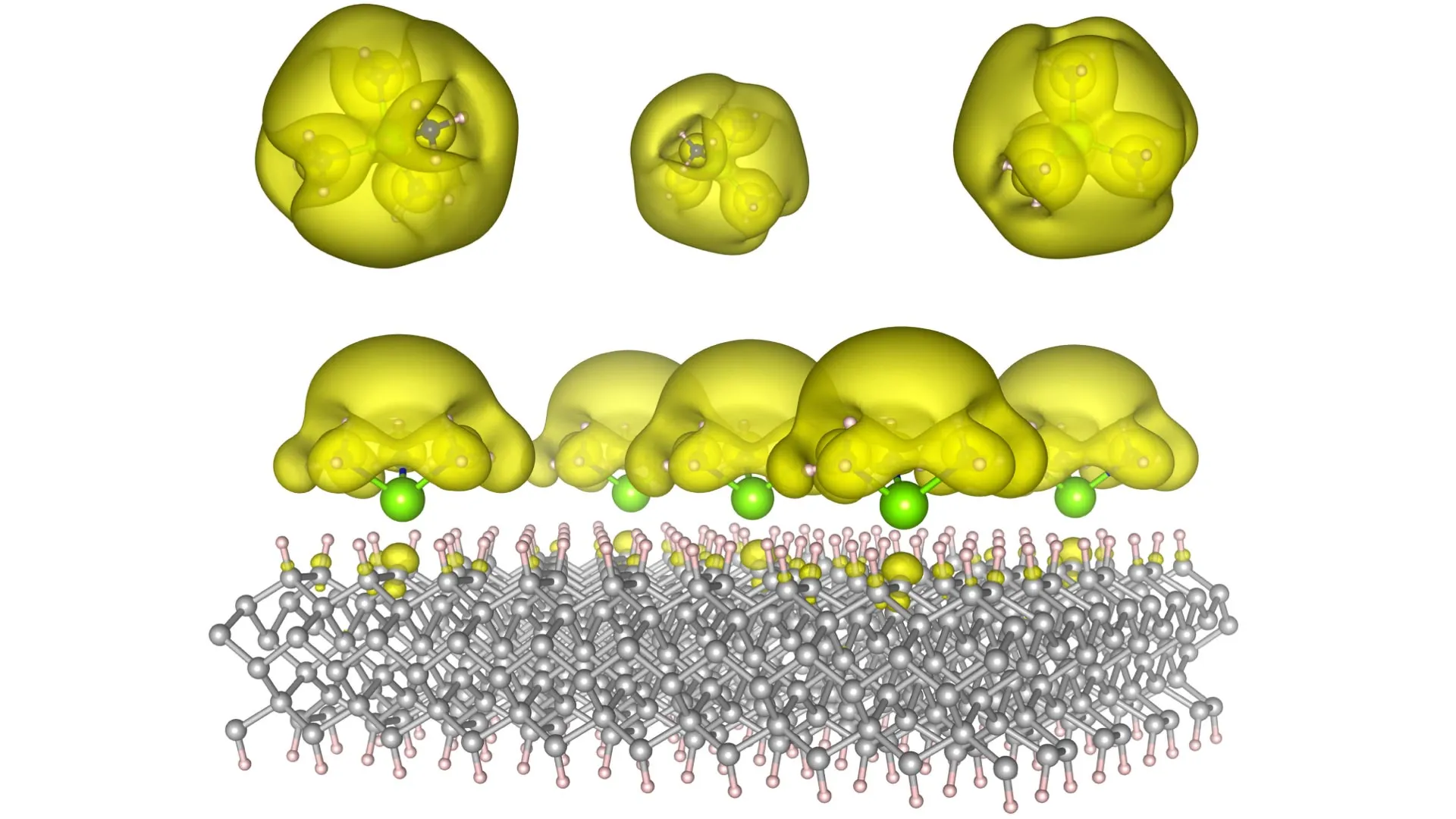
Electrons, these ubiquitous and fundamental constituents of matter, are the very linchpins of nearly every chemical transformation and technological innovation that shapes our modern world. They are the silent conductors of energy transfer, the architects of chemical bonds that hold matter together, and the indispensable carriers of electrical current. In essence, they form the bedrock upon which both the intricate art of chemical synthesis and the sophisticated realm of modern electronics are built. Within the dynamic arena of chemical reactions, electrons are the active agents enabling vital redox processes, facilitating the formation of new molecular bonds, and acting as potent catalysts that accelerate reaction rates. In the technological sphere, the artful management of electron movement and interaction is the silent engine powering everything from the microscopic transistors in our electronic circuits and the sophisticated algorithms driving artificial intelligence systems, to the photovoltaic conversion in solar cells and the mind-bending potential of quantum computers. Traditionally, electrons are largely confined to the atomic orbits of individual atoms, a confinement that inherently limits their potential for novel applications and functionalities. However, a unique class of materials known as electrides represents a radical departure from this norm. In electrides, electrons exist in a state of liberation, moving independently and unbound, thus unlocking a vista of remarkable new capabilities that were previously unimaginable.
Dr. Evangelos Miliordos, an Associate Professor of Chemistry at Auburn University and the senior architect behind this transformative study, eloquently articulates the profound implications of this discovery: "By acquiring the profound knowledge and capability to precisely control these free-roaming electrons, we are empowered to design and engineer materials that can perform functions and exhibit properties far beyond what nature has conventionally intended or allowed." The groundbreaking research that underpins this assertion was meticulously conducted, leveraging the power of advanced computational modeling to explore and predict the behavior of these novel electronic states.
In their pursuit of this unprecedented electron control, the Auburn research team ingeniously devised innovative material architectures, christened "Surface Immobilized Electrides." This revolutionary approach involves the strategic attachment of solvated electron precursors to highly stable and robust solid surfaces, such as diamond and silicon carbide. This deliberate architectural design imbues the electrides with a dual advantage: their electronic characteristics are rendered both remarkably durable, capable of withstanding the rigors of practical application, and exquisitely tunable, allowing for precise modification of their electronic behavior. The researchers discovered that by subtly altering the spatial arrangement and molecular architecture of these precursors on the surface, they could dictate the behavior of the electrons. They can be coaxed to cluster into isolated, discrete "islands," each acting as a self-contained quantum unit, which holds immense promise for the development of advanced computing paradigms, particularly in the realm of quantum computing. Alternatively, these electrons can be encouraged to spread out and delocalize into extended, interconnected "seas," a configuration that is exceptionally conducive to promoting and accelerating complex chemical reactions, opening new avenues for catalytic innovation.
This inherent versatility is precisely what endows this remarkable discovery with its truly transformative potential. One manifestation of this research could pave the way for the creation of immensely powerful quantum computers, machines capable of tackling and solving problems that currently lie far beyond the computational reach of even the most advanced supercomputers available today. Concurrently, another application of these electrides could form the very foundation for a new generation of cutting-edge catalysts. These novel catalysts would possess the remarkable ability to dramatically accelerate essential chemical reactions, thereby revolutionizing the way we produce everything from critical fuels and life-saving pharmaceuticals to a vast array of essential industrial materials, ushering in an era of greener and more efficient manufacturing.
Dr. Marcelo Kuroda, another Associate Professor of Physics at Auburn and a key contributor to the study, emphasizes the pressing need for such advanced materials: "As our global society relentlessly pushes the boundaries of current technological capabilities, the demand for entirely new classes of materials with unprecedented properties is experiencing an exponential surge. Our pioneering work illuminates a novel and promising pathway towards the creation of materials that not only offer unparalleled opportunities for fundamental scientific investigations into the intricate interactions within matter but also hold immense promise for a wide spectrum of practical, real-world applications."
Previous attempts to synthesize and utilize electrides were often hampered by their inherent instability and the formidable challenges associated with scaling up their production. By ingeniously depositing these electrides directly onto solid surfaces, the Auburn team has successfully circumvented these critical barriers. They have proposed a versatile and robust family of material structures that are poised to transition from the theoretical constructs of computational models to tangible, functional devices that can be integrated into real-world technologies. Dr. Konstantin Klyukin, an Assistant Professor of Materials Engineering at Auburn and a co-author of the study, underscores the profound real-world impact of this fundamental scientific breakthrough: "This is undeniably fundamental science at its core, yet its implications are remarkably tangible and far-reaching. We are envisioning and actively working towards technologies that possess the potential to fundamentally alter the way we compute and the way we manufacture goods, leading to paradigm shifts in both domains."
The theoretical underpinnings of this groundbreaking study were meticulously developed and orchestrated by a multidisciplinary team of esteemed faculty members spanning the departments of Chemistry, Physics, and Materials Engineering at Auburn University. Dr. Miliordos concludes with a forward-looking perspective, stating, "This monumental discovery represents merely the nascent beginnings of an exciting new frontier. By diligently learning and mastering the art of taming and controlling free electrons, we can now vividly imagine a future replete with computers that operate at speeds we can scarcely comprehend, machines that exhibit unprecedented levels of intelligence and adaptability, and the emergence of entirely new technologies that, at this very moment, we haven’t even begun to dream of."
The comprehensive study, bearing the title "Electrides with Tunable Electron Delocalization for Applications in Quantum Computing and Catalysis," was further enriched by the invaluable contributions of graduate students Andrei Evdokimov and Valentina Nesterova, who played integral roles in the research and data analysis. The endeavor received crucial support from the U.S. National Science Foundation, a testament to its scientific merit and potential impact, as well as essential computational resources provided by Auburn University, which were indispensable for the sophisticated modeling and simulations undertaken.