Electrons, the fundamental carriers of negative charge, are the unsung heroes behind virtually every chemical reaction and technological advancement. Their intricate dance dictates energy transfer, the formation of chemical bonds, and the flow of electricity, forming the bedrock of both sophisticated chemical synthesis and the ubiquitous digital landscape of modern electronics. In the realm of chemistry, electrons are the arbiters of redox processes, the architects of molecular connections, and the catalysts that accelerate vital reactions. In the technological sphere, the ability to manipulate electron movement and interaction underpins the functionality of everything from the microprocessors in our devices and the complex algorithms driving artificial intelligence, to the energy conversion mechanisms in solar cells and the mind-bending computations of quantum computers. Traditionally, electrons have been largely confined to the atomic orbitals of specific atoms, a limitation that has, until now, constrained their full potential. However, a special class of materials known as electrides offers a tantalizing glimpse into a future where electrons are liberated, moving independently and paving the way for an array of extraordinary new capabilities.
"By mastering the art of controlling these free-roaming electrons, we are empowered to design and create materials that can perform functions far beyond anything observed in nature," explains Dr. Evangelos Miliordos, an Associate Professor of Chemistry at Auburn University and the senior author of this pivotal study, which was extensively informed by advanced computational modeling. This profound insight underscores the transformative nature of the Auburn team’s research, moving beyond observation to active design and engineering of matter at its most fundamental level.
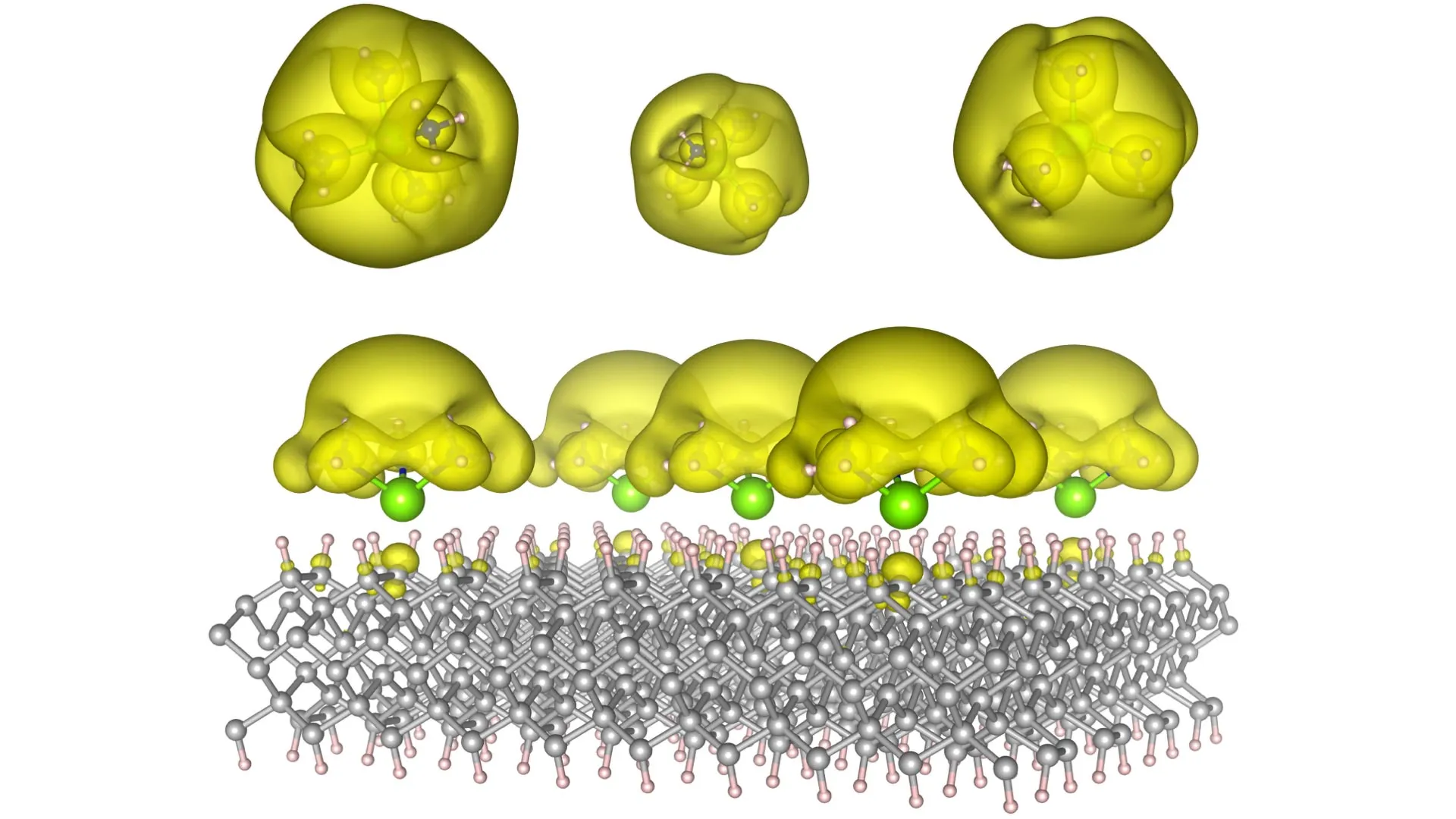
The ingenuity of the Auburn team lies in their novel approach to material fabrication. They have pioneered the creation of unique material structures termed "Surface Immobilized Electrides." This is achieved by meticulously anchoring solvated electron precursors onto robust and stable surfaces, such as diamond and silicon carbide. This strategic integration imbues the electrides with a remarkable degree of durability and, crucially, tunability. By precisely orchestrating the arrangement of these molecular complexes, the researchers can dictate the behavior of the electrons. They can induce electrons to coalesce into discrete, isolated "islands," a configuration that mimics the behavior of quantum bits, or qubits, the fundamental units of information in quantum computing. Alternatively, they can encourage electrons to spread out into vast, interconnected "seas," a state that significantly enhances their capacity to facilitate and accelerate complex chemical reactions.
This inherent versatility is the very essence of the discovery’s transformative potential. One manifestation of these new materials could be the development of vastly more powerful quantum computers. These machines would possess the computational prowess to tackle problems that are currently intractable for even the most advanced supercomputers, potentially revolutionizing fields like drug discovery, materials science, and complex system modeling. Concurrently, another configuration of these electrides could serve as the foundation for a new generation of cutting-edge catalysts. These catalysts would dramatically accelerate essential chemical reactions, promising to fundamentally reshape the production of everything from clean fuels and life-saving pharmaceuticals to the vast array of industrial materials that underpin modern society.
"As our global society relentlessly pushes the boundaries of existing technological capabilities, the demand for novel and advanced materials is experiencing an exponential surge," observes Dr. Marcelo Kuroda, an Associate Professor of Physics at Auburn University. "Our research illuminates a promising new pathway towards materials that not only offer unprecedented opportunities for fundamental scientific investigations into the intricate interactions within matter but also hold immense promise for practical, real-world applications." This sentiment highlights the dual impact of the discovery, bridging the gap between abstract scientific inquiry and tangible technological advancement.
Previous attempts to harness the power of electrides were often hampered by their inherent instability and the significant challenges associated with scaling up their production. The Auburn team’s innovative strategy of depositing these electrides directly onto solid surfaces has effectively circumvented these long-standing barriers. This breakthrough paves the way for a robust family of material structures that can transition from theoretical constructs and laboratory curiosities to fully realized, functional devices. "While this represents fundamental scientific research, its implications are profoundly real and far-reaching," emphasizes Dr. Konstantin Klyukin, an Assistant Professor of Materials Engineering at Auburn University. "We are envisioning technologies that have the potential to fundamentally alter the very fabric of how we compute and how we manufacture goods."
The intellectual genesis of this pioneering theoretical study spanned across multiple disciplines within Auburn University, drawing expertise from the departments of Chemistry, Physics, and Materials Engineering. This interdisciplinary approach was crucial in addressing the multifaceted challenges inherent in controlling electron behavior at such a fundamental level. "This is merely the nascent stage of what we believe will be a monumental journey," Dr. Miliordos adds with evident enthusiasm. "By unraveling the secrets of how to effectively tame and manipulate free electrons, we can begin to conceptualize and engineer a future characterized by dramatically faster computing, more intelligent machines, and entirely new technological paradigms that we have yet to even imagine." The study, formally titled "Electrides with Tunable Electron Delocalization for Applications in Quantum Computing and Catalysis," was further enriched by the significant contributions of graduate students Andrei Evdokimov and Valentina Nesterova, whose dedicated efforts were instrumental in bringing this complex research to fruition. The foundational work was generously supported by the U.S. National Science Foundation and the extensive computing resources provided by Auburn University, underscoring the collaborative and resource-intensive nature of modern scientific discovery.